Main Menu (Mobile)- Block
- Our Research
-
Support Teams
- Overview
- Anatomy and Histology
- Cryo-Electron Microscopy
- Electron Microscopy
- Flow Cytometry
- Fly Facility
- Gene Targeting and Transgenics
- Immortalized Cell Line Culture
- Integrative Imaging
- Janelia Experimental Technology
- Media Prep
- Molecular Genomics
- Primary & iPS Cell Culture
- Project Pipeline Support
- Project Technical Resources
- Quantitative Genomics
- Scientific Computing Software
- Scientific Computing Systems
- Viral Tools
- Vivarium
- Open Science
- You + Janelia
- About Us
Main Menu - Block
- Overview
- Anatomy and Histology
- Cryo-Electron Microscopy
- Electron Microscopy
- Flow Cytometry
- Fly Facility
- Gene Targeting and Transgenics
- Immortalized Cell Line Culture
- Integrative Imaging
- Janelia Experimental Technology
- Media Prep
- Molecular Genomics
- Primary & iPS Cell Culture
- Project Pipeline Support
- Project Technical Resources
- Quantitative Genomics
- Scientific Computing Software
- Scientific Computing Systems
- Viral Tools
- Vivarium
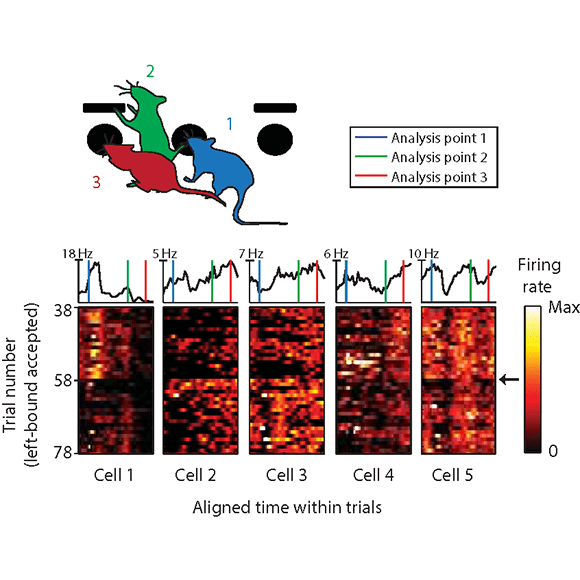
We are interested in understanding what neural computations allow animals to dynamically adjust their behavior according to the structure and demands of the environment and how these circuit mechanisms are perturbed in neuropsychiatric disorders.
In order for animals to survive in complex, natural and ever-changing environments they must be able to make inferences about the world on the basis of sparse and often ambiguous data.
One way to effectively navigate a complex environment is by attempting to construct an internal model of the environment’s governing rules. Such an internal model not only computes an estimate of the current environmental state based on available data, but discovers and incorporates general principles about the causal rules that operate in the environment. The overall interest of our lab is to understand how model-based inference is accomplished by neural circuits. Causal reasoning and the ability to acquire abstract hierarchical relational schemas has been shown for rats, making them a good model for uncovering the underlying circuit computations. Our approach thus relies on a combination of behavioral tasks in rats designed to isolate specific aspects of model acquisition and updating, recordings of the underlying neural ensemble dynamics, perturbation of specific circuit elements and computational modeling.
