The activity of principal neurons in the hippocampus depends on the position of an animal in the environment - different principal neurons are active when an animal is in different locations in its environment, as if activity of a specific neuron enables an animal to know where it is (Fig. 1.). That is why these neurons are named “place cells”.
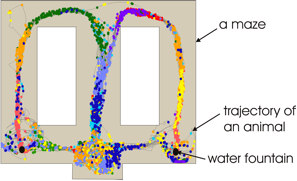
Activity of hippocampal place cells depends on the position of an animal in its environment. Dots of the same color represent the position of an animal when one place cell was activated. Different colors represent the activity of different place cells. Activity of different place cells varies: the size and the number of the areas in which cells are active might differ between different cells, different cells might have different level of activation, etc. However, the same neurons are activated in a very similar manner anytime the same animal enters the same environment.
We found that some principal neurons that we dubbed “episode cells” are active in a very similar manner when an animal stops its motion through the environment while it is engaged in a memory task - different episode cells are active at different time points after the animal has stopped its motion (Fig. 2.). This indicates that the activity of a particular neuron informs an animal that it is in a specific part of its thought/memory/plan/episode. That is why we called these neurons “episode cells”.
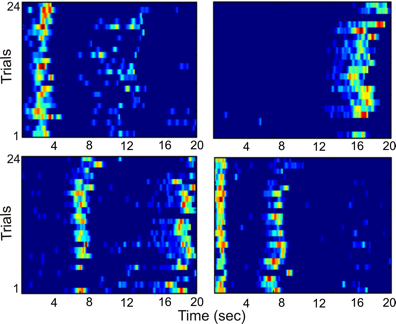
Activity of four episode cells in time since an animal stopped its motion through the environment during a memory task. one square represents the activity of one episode cell color represents the level of the activation of an episode cell (blue – no activity, yellow – medium level of activity, red – high level of activity) the x axis represents the time since the animal stopped its motion through the environment, the y axis represents repeated trials.
It is important to note that activity of hippocampal principal neurons is very different when an animal becomes stationary while it is NOT engaged in a memory task - none of the neurons is activated at a specific time once the animal stops moving. Neurons are activated in a random manner with respect to the beginning of the stationary period (Fig. 3.).
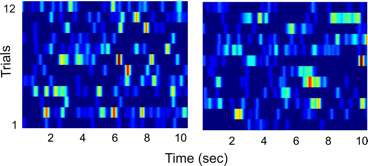
Activity of two hippocampal neurons in time since an animal stopped its motion through the environment while it was NOT involved in a memory task. one square represents the activity of one principal hippocampal neuron color represents the level of the activation of a neuron (blue – no activity, yellow – medium level of activity, red – high level of activity) the x axis represents the time since the animal stopped its motion through the environment the y axis represents repeated trials.
Now we know that the hippocampal network can operate in two different modes and that the ‘choice’ of the mode depends on whether the animal is involved in a memory task. As if an animal imagines its future trajectory/sequence of actions while it is stationary during the memory task but does not imagine anything specific while it is stationary during a non-memory task. We are now studying how these two modes of activity are generated. Specifically, we are asking whether these two modes are caused by:
- different inputs into the hippocampal network
- different interactions between local hippocampal neurons
- different timing between hippocampal inputs and the local activity
