Filter
Associated Lab
- Aguilera Castrejon Lab (17) Apply Aguilera Castrejon Lab filter
- Ahrens Lab (68) Apply Ahrens Lab filter
- Aso Lab (42) Apply Aso Lab filter
- Baker Lab (38) Apply Baker Lab filter
- Betzig Lab (115) Apply Betzig Lab filter
- Beyene Lab (14) Apply Beyene Lab filter
- Bock Lab (17) Apply Bock Lab filter
- Branson Lab (54) Apply Branson Lab filter
- Card Lab (43) Apply Card Lab filter
- Cardona Lab (64) Apply Cardona Lab filter
- Chklovskii Lab (13) Apply Chklovskii Lab filter
- Clapham Lab (15) Apply Clapham Lab filter
- Cui Lab (19) Apply Cui Lab filter
- Darshan Lab (12) Apply Darshan Lab filter
- Dennis Lab (1) Apply Dennis Lab filter
- Dickson Lab (46) Apply Dickson Lab filter
- Druckmann Lab (25) Apply Druckmann Lab filter
- Dudman Lab (52) Apply Dudman Lab filter
- Eddy/Rivas Lab (30) Apply Eddy/Rivas Lab filter
- Egnor Lab (11) Apply Egnor Lab filter
- Espinosa Medina Lab (20) Apply Espinosa Medina Lab filter
- Feliciano Lab (9) Apply Feliciano Lab filter
- Fetter Lab (41) Apply Fetter Lab filter
- FIB-SEM Technology (1) Apply FIB-SEM Technology filter
- Fitzgerald Lab (29) Apply Fitzgerald Lab filter
- Freeman Lab (15) Apply Freeman Lab filter
- Funke Lab (41) Apply Funke Lab filter
- Gonen Lab (91) Apply Gonen Lab filter
- Grigorieff Lab (62) Apply Grigorieff Lab filter
- Harris Lab (64) Apply Harris Lab filter
- Heberlein Lab (94) Apply Heberlein Lab filter
- Hermundstad Lab (29) Apply Hermundstad Lab filter
- Hess Lab (79) Apply Hess Lab filter
- Ilanges Lab (2) Apply Ilanges Lab filter
- Jayaraman Lab (47) Apply Jayaraman Lab filter
- Ji Lab (33) Apply Ji Lab filter
- Johnson Lab (6) Apply Johnson Lab filter
- Kainmueller Lab (19) Apply Kainmueller Lab filter
- Karpova Lab (14) Apply Karpova Lab filter
- Keleman Lab (13) Apply Keleman Lab filter
- Keller Lab (76) Apply Keller Lab filter
- Koay Lab (18) Apply Koay Lab filter
- Lavis Lab (153) Apply Lavis Lab filter
- Lee (Albert) Lab (34) Apply Lee (Albert) Lab filter
- Leonardo Lab (23) Apply Leonardo Lab filter
- Li Lab (30) Apply Li Lab filter
- Lippincott-Schwartz Lab (177) Apply Lippincott-Schwartz Lab filter
- Liu (Yin) Lab (7) Apply Liu (Yin) Lab filter
- Liu (Zhe) Lab (64) Apply Liu (Zhe) Lab filter
- Looger Lab (138) Apply Looger Lab filter
- Magee Lab (49) Apply Magee Lab filter
- Menon Lab (18) Apply Menon Lab filter
- Murphy Lab (13) Apply Murphy Lab filter
- O'Shea Lab (7) Apply O'Shea Lab filter
- Otopalik Lab (13) Apply Otopalik Lab filter
- Pachitariu Lab (49) Apply Pachitariu Lab filter
- Pastalkova Lab (18) Apply Pastalkova Lab filter
- Pavlopoulos Lab (19) Apply Pavlopoulos Lab filter
- Pedram Lab (15) Apply Pedram Lab filter
- Podgorski Lab (16) Apply Podgorski Lab filter
- Reiser Lab (52) Apply Reiser Lab filter
- Riddiford Lab (44) Apply Riddiford Lab filter
- Romani Lab (48) Apply Romani Lab filter
- Rubin Lab (147) Apply Rubin Lab filter
- Saalfeld Lab (64) Apply Saalfeld Lab filter
- Satou Lab (16) Apply Satou Lab filter
- Scheffer Lab (38) Apply Scheffer Lab filter
- Schreiter Lab (68) Apply Schreiter Lab filter
- Sgro Lab (21) Apply Sgro Lab filter
- Shroff Lab (31) Apply Shroff Lab filter
- Simpson Lab (23) Apply Simpson Lab filter
- Singer Lab (80) Apply Singer Lab filter
- Spruston Lab (95) Apply Spruston Lab filter
- Stern Lab (158) Apply Stern Lab filter
- Sternson Lab (54) Apply Sternson Lab filter
- Stringer Lab (39) Apply Stringer Lab filter
- Svoboda Lab (135) Apply Svoboda Lab filter
- Tebo Lab (35) Apply Tebo Lab filter
- Tervo Lab (9) Apply Tervo Lab filter
- Tillberg Lab (21) Apply Tillberg Lab filter
- Tjian Lab (64) Apply Tjian Lab filter
- Truman Lab (88) Apply Truman Lab filter
- Turaga Lab (53) Apply Turaga Lab filter
- Turner Lab (39) Apply Turner Lab filter
- Vale Lab (8) Apply Vale Lab filter
- Voigts Lab (3) Apply Voigts Lab filter
- Wang (Meng) Lab (24) Apply Wang (Meng) Lab filter
- Wang (Shaohe) Lab (25) Apply Wang (Shaohe) Lab filter
- Wu Lab (9) Apply Wu Lab filter
- Zlatic Lab (28) Apply Zlatic Lab filter
- Zuker Lab (25) Apply Zuker Lab filter
Associated Project Team
- CellMap (12) Apply CellMap filter
- COSEM (3) Apply COSEM filter
- FIB-SEM Technology (5) Apply FIB-SEM Technology filter
- Fly Descending Interneuron (12) Apply Fly Descending Interneuron filter
- Fly Functional Connectome (14) Apply Fly Functional Connectome filter
- Fly Olympiad (5) Apply Fly Olympiad filter
- FlyEM (56) Apply FlyEM filter
- FlyLight (50) Apply FlyLight filter
- GENIE (47) Apply GENIE filter
- Integrative Imaging (6) Apply Integrative Imaging filter
- Larval Olympiad (2) Apply Larval Olympiad filter
- MouseLight (18) Apply MouseLight filter
- NeuroSeq (1) Apply NeuroSeq filter
- ThalamoSeq (1) Apply ThalamoSeq filter
- Tool Translation Team (T3) (28) Apply Tool Translation Team (T3) filter
- Transcription Imaging (49) Apply Transcription Imaging filter
Publication Date
- 2025 (203) Apply 2025 filter
- 2024 (212) Apply 2024 filter
- 2023 (159) Apply 2023 filter
- 2022 (192) Apply 2022 filter
- 2021 (194) Apply 2021 filter
- 2020 (196) Apply 2020 filter
- 2019 (202) Apply 2019 filter
- 2018 (232) Apply 2018 filter
- 2017 (217) Apply 2017 filter
- 2016 (209) Apply 2016 filter
- 2015 (252) Apply 2015 filter
- 2014 (236) Apply 2014 filter
- 2013 (194) Apply 2013 filter
- 2012 (190) Apply 2012 filter
- 2011 (190) Apply 2011 filter
- 2010 (161) Apply 2010 filter
- 2009 (158) Apply 2009 filter
- 2008 (140) Apply 2008 filter
- 2007 (106) Apply 2007 filter
- 2006 (92) Apply 2006 filter
- 2005 (67) Apply 2005 filter
- 2004 (57) Apply 2004 filter
- 2003 (58) Apply 2003 filter
- 2002 (39) Apply 2002 filter
- 2001 (28) Apply 2001 filter
- 2000 (29) Apply 2000 filter
- 1999 (14) Apply 1999 filter
- 1998 (18) Apply 1998 filter
- 1997 (16) Apply 1997 filter
- 1996 (10) Apply 1996 filter
- 1995 (18) Apply 1995 filter
- 1994 (12) Apply 1994 filter
- 1993 (10) Apply 1993 filter
- 1992 (6) Apply 1992 filter
- 1991 (11) Apply 1991 filter
- 1990 (11) Apply 1990 filter
- 1989 (6) Apply 1989 filter
- 1988 (1) Apply 1988 filter
- 1987 (7) Apply 1987 filter
- 1986 (4) Apply 1986 filter
- 1985 (5) Apply 1985 filter
- 1984 (2) Apply 1984 filter
- 1983 (2) Apply 1983 filter
- 1982 (3) Apply 1982 filter
- 1981 (3) Apply 1981 filter
- 1980 (1) Apply 1980 filter
- 1979 (1) Apply 1979 filter
- 1976 (2) Apply 1976 filter
- 1973 (1) Apply 1973 filter
- 1970 (1) Apply 1970 filter
- 1967 (1) Apply 1967 filter
Type of Publication
4179 Publications
Showing 861-870 of 4179 resultsPreclinical animal models have provided strong evidence that estrogen (E) therapy (ET) enhances cognition and induces spinogenesis in neuronal circuits. However, clinical studies have been inconsistent, with some studies revealing adverse effects of ET, including an increased risk of dementia. In an effort to bridge this disconnect between the preclinical and clinical data, we have developed a nonhuman primate (NHP) model of ET combined with high-resolution dendritic spine analysis of dorsolateral prefrontal cortical (dlPFC) neurons. Previously, we reported cyclic ET in aged, ovariectomized NHPs increased spine density on dlPFC neurons. Here, we report that monkeys treated with cyclic E treatment paired with cyclic progesterone (P), continuous E combined with P (either cyclic or continuous), or unopposed continuous E failed to increase spines on dlPFC neurons. Given that the most prevalent form of ET prescribed to women is a combined and continuous E and P, these data bring into convergence the human neuropsychological findings and preclinical neurobiological evidence that standard hormone therapy in women is unlikely to yield the synaptic benefit presumed to underlie the cognitive enhancement reported in animal models.
Detecting meaningful structure in neural activity and connectivity data is challenging in the presence of hidden nonlinearities, where traditional eigenvalue-based methods may be misleading. We introduce a novel approach to matrix analysis, called clique topology, that extracts features of the data invariant under nonlinear monotone transformations. These features can be used to detect both random and geometric structure, and depend only on the relative ordering of matrix entries. We then analyzed the activity of pyramidal neurons in rat hippocampus, recorded while the animal was exploring a 2D environment, and confirmed that our method is able to detect geometric organization using only the intrinsic pattern of neural correlations. Remarkably, we found similar results during nonspatial behaviors such as wheel running and rapid eye movement (REM) sleep. This suggests that the geometric structure of correlations is shaped by the underlying hippocampal circuits and is not merely a consequence of position coding. We propose that clique topology is a powerful new tool for matrix analysis in biological settings, where the relationship of observed quantities to more meaningful variables is often nonlinear and unknown.
The antennal lobe (AL) is the primary structure in the Drosophila brain that relays odor information from the antennae to higher brain centers. The characterization of uniglomerular projection neurons (PNs) and some local interneurons has facilitated our understanding of olfaction; however, many other AL neurons remain unidentified. Because neuron types are mostly specified by lineage and temporal origins, we use the MARCM techniques with a set of enhancer-trap GAL4 lines to perform systematical lineage analysis to characterize neuron morphologies, lineage origin and birth timing in the three AL neuron lineages that contain GAL4-GH146-positive PNs: anterodorsal, lateral and ventral lineages. The results show that the anterodorsal lineage is composed of pure uniglomerular PNs that project through the inner antennocerebral tract. The ventral lineage produces uniglomerular and multiglomerular PNs that project through the middle antennocerebral tract. The lateral lineage generates multiple types of neurons, including uniglomeurlar PNs, diverse atypical PNs, various types of AL local interneurons and the neurons that make no connection within the ALs. Specific neuron types in all three lineages are produced in specific time windows, although multiple neuron types in the lateral lineage are made simultaneously. These systematic cell lineage analyses have not only filled gaps in the olfactory map, but have also exemplified additional strategies used in the brain to increase neuronal diversity.
BACKGROUND: The insect brain can be divided into neuropils that are formed by neurites of both local and remote origin. The complexity of the interconnections obscures how these neuropils are established and interconnected through development. The Drosophila central brain develops from a fixed number of neuroblasts (NBs) that deposit neurons in regional clusters. RESULTS: By determining individual NB clones and pursuing their projections into specific neuropils, we unravel the regional development of the brain neural network. Exhaustive clonal analysis revealed 95 stereotyped neuronal lineages with characteristic cell-body locations and neurite trajectories. Most clones show complex projection patterns, but despite the complexity, neighboring clones often coinnervate the same local neuropil or neuropils and further target a restricted set of distant neuropils. CONCLUSIONS: These observations argue for regional clonal development of both neuropils and neuropil connectivity throughout the Drosophila central brain.
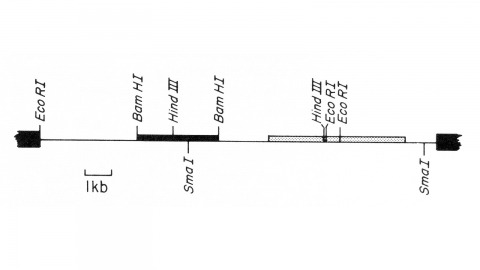
View Publication PageWe describe the isolation of a cloned DNA segment carrying unique sequences from the white locus of Drosophila melanogaster. Sequences within the cloned segment are shown to hybridize in situ to the white locus region on the polytene chromosomes of both wild-type strains and strains carrying chromosomal rearrangements whose breakpoints bracket the white locus. We further show that two small deficiency mutations, deleting white locus genetic elements but not those of complementation groups contiguous to white, delete the genomic sequences corresponding to a portion of the cloned segment. The strategy we have employed to isolate this cloned segment exploits the existence of an allele at the white locus containing a copy of a previously cloned transposable, reiterated DNA sequence element. We describe a simple, rapid method for retrieving cloned segments carrying a copy of the transposable element together with contiguous sequences corresponding to this allele. The strategy described is potentially general and we discuss its application to the cloning of the DNA sequences of other genes in Drosophila, including those identified only by genetic analysis and for which no RNA product is known.
Multipath propagation in shallow water can lead to mismatch losses when single-path replicas are usedfor horizontal array beamforming.Matched field processing(MFP) seeks to remedy this by using full-fieldacoustic propagationmodels to predict the multipath arrival structure. Ideally MFP can give source localization in range and depth as well as detection gains but robustly estimating range and depth is difficult in practice. The approach described here seeks to collapse full-field replica outputs to bearing which is robustly estimated while retaining any signal gains provided by the full-field model.Clusteranalysis is used to group together full-field replicas with similar responses. This yields a less redundant “sampled field” describing a set of representative multipath structures for each bearing. A detection algorithm is introduced that uses clustering to collapse beamformer outputs to bearing such that signal gains are retained while increases in the noise floor are minimized. Horizontal array data from SWELLEX-96 are used to demonstrate the detection benefits of sampled field as compared to single-pathbeamforming.
Advances in single-cell RNA-sequencing technology have resulted in a wealth of studies aiming to identify transcriptomic cell types in various biological systems. There are multiple experimental approaches to isolate and profile single cells, which provide different levels of cellular and tissue coverage. In addition, multiple computational strategies have been proposed to identify putative cell types from single-cell data. From a data generation perspective, recent single-cell studies can be classified into two groups: those that distribute reads shallowly over large numbers of cells and those that distribute reads more deeply over a smaller cell population. Although there are advantages to both approaches in terms of cellular and tissue coverage, it is unclear whether different computational cell type identification methods are better suited to one or the other experimental paradigm. This study reviews three cell type clustering algorithms, each representing one of three broad approaches, and finds that PCA-based algorithms appear most suited to low read depth data sets, whereas gene clustering-based and biclustering algorithms perform better on high read depth data sets. In addition, highly related cell classes are better distinguished by higher-depth data, given the same total number of reads; however, simultaneous discovery of distinct and similar types is better served by lower-depth, higher cell number data. Overall, this study suggests that the depth of profiling should be determined by initial assumptions about the diversity of cells in the population, and that the selection of clustering algorithm(s) is subsequently based on the depth of profiling will allow for better identification of putative transcriptomic cell types.
The evolution of sexual traits often involves correlated changes in morphology and behavior. For example, in Drosophila, divergent mating displays are often accompanied by divergent pigment patterns. To better understand how such traits co-evolve, we investigated the genetic basis of correlated divergence in wing pigmentation and mating display between the sibling species Drosophila elegans and D. gunungcola. Drosophila elegans males have an area of black pigment on their wings known as a wing spot and appear to display this spot to females by extending their wings laterally during courtship. By contrast, D. gunungcola lost both of these traits. Using Multiplexed Shotgun Genotyping (MSG), we identified a ∼440 kb region on the X chromosome that behaves like a genetic switch controlling the presence or absence of male-specific wing spots. This region includes the candidate gene optomotor-blind (omb), which plays a critical role in patterning the Drosophila wing. The genetic basis of divergent wing display is more complex, with at least two loci on the X chromosome and two loci on autosomes contributing to its evolution. Introgressing the X-linked region affecting wing spot development from D. gunungcola into D. elegans reduced pigmentation in the wing spots but did not affect the wing display, indicating that these are genetically separable traits. Consistent with this observation, broader sampling of wild D. gunungcola populations confirmed the wing spot and wing display are evolving independently: some D. gunungcola males performed wing displays similar to D. elegans despite lacking wing spots. These data suggest that correlated selection pressures rather than physical linkage or pleiotropy are responsible for the coevolution of these morphological and behavioral traits. They also suggest that the change in morphology evolved prior to the change in behavior. This article is protected by copyright. All rights reserved.
Cells often fine-tune gene expression at the level of transcription to generate the appropriate response to a given environmental or developmental stimulus. Both positive and negative influences on gene expression must be balanced to produce the correct level of mRNA synthesis. To this end, the cell uses several classes of regulatory coactivator complexes including two central players, TFIID and Mediator (MED), in potentiating activated transcription. Both of these complexes integrate activator signals and convey them to the basal apparatus. Interestingly, many promoters require both regulatory complexes, although at first glance they may seem to be redundant. Here we have used RNA interference (RNAi) in Drosophila cells to selectively deplete subunits of the MED and TFIID complexes to dissect the contribution of each of these complexes in modulating activated transcription. We exploited the robust response of the metallothionein genes to heavy metal as a model for transcriptional activation by analyzing direct factor recruitment in both heterogeneous cell populations and at the single-cell level. Intriguingly, we find that MED and TFIID interact functionally to modulate transcriptional response to metal. The metal response element-binding transcription factor-1 (MTF-1) recruits TFIID, which then binds promoter DNA, setting up a "checkpoint complex" for the initiation of transcription that is subsequently activated upon recruitment of the MED complex. The appropriate expression level of the endogenous metallothionein genes is achieved only when the activities of these two coactivators are balanced. Surprisingly, we find that the same activator (MTF-1) requires different coactivator subunits depending on the context of the core promoter. Finally, we find that the stability of multi-subunit coactivator complexes can be compromised by loss of a single subunit, underscoring the potential for combinatorial control of transcription activation.