Filter
Associated Lab
- Ahrens Lab (4) Apply Ahrens Lab filter
- Betzig Lab (1) Apply Betzig Lab filter
- Beyene Lab (1) Apply Beyene Lab filter
- Druckmann Lab (1) Apply Druckmann Lab filter
- Harris Lab (4) Apply Harris Lab filter
- Hermundstad Lab (1) Apply Hermundstad Lab filter
- Jayaraman Lab (9) Apply Jayaraman Lab filter
- Karpova Lab (1) Apply Karpova Lab filter
- Keller Lab (1) Apply Keller Lab filter
- Lavis Lab (8) Apply Lavis Lab filter
- Leonardo Lab (1) Apply Leonardo Lab filter
- Liu (Zhe) Lab (1) Apply Liu (Zhe) Lab filter
- Looger Lab (24) Apply Looger Lab filter
- Podgorski Lab (5) Apply Podgorski Lab filter
- Rubin Lab (1) Apply Rubin Lab filter
- Schreiter Lab (70) Apply Schreiter Lab filter
- Stringer Lab (1) Apply Stringer Lab filter
- Svoboda Lab (13) Apply Svoboda Lab filter
- Tillberg Lab (1) Apply Tillberg Lab filter
- Turner Lab (4) Apply Turner Lab filter
- Zlatic Lab (1) Apply Zlatic Lab filter
Associated Project Team
Publication Date
- 2026 (1) Apply 2026 filter
- 2025 (5) Apply 2025 filter
- 2024 (5) Apply 2024 filter
- 2023 (6) Apply 2023 filter
- 2021 (1) Apply 2021 filter
- 2020 (5) Apply 2020 filter
- 2019 (4) Apply 2019 filter
- 2018 (4) Apply 2018 filter
- 2017 (4) Apply 2017 filter
- 2016 (2) Apply 2016 filter
- 2015 (4) Apply 2015 filter
- 2013 (7) Apply 2013 filter
- 2012 (2) Apply 2012 filter
- 2011 (3) Apply 2011 filter
- 2010 (2) Apply 2010 filter
- 2009 (3) Apply 2009 filter
- 2008 (3) Apply 2008 filter
- 2007 (3) Apply 2007 filter
- 2006 (2) Apply 2006 filter
- 2003 (1) Apply 2003 filter
- 2001 (2) Apply 2001 filter
- 1999 (1) Apply 1999 filter
Type of Publication
70 Publications
Showing 41-50 of 70 resultsWe have developed a series of yellow genetically encoded Ca indicators for optical imaging (Y-GECOs) with inverted responses to Ca and apparent dissociation constants (K') ranging from 25 to 2400 nM. To demonstrate the utility of this affinity series of Ca indicators, we expressed the four highest affinity variants (K's = 25, 63, 121, and 190 nM) in the Drosophila medulla intrinsic neuron Mi1. Hyperpolarization of Mi1 by optogenetic stimulation of the laminar monopolar neuron L1 produced a decrease in intracellular Ca in layers 8-10, and a corresponding increase in Y-GECO fluorescence. These experiments revealed that lower K' was associated with greater increases in fluorescence, but longer delays to reach the maximum signal change due to slower off-rate kinetics.
We have prepared ionic liquids by mixing either iron(II) chloride or iron(III) chloride with 1-butyl-3-methylimidazolium chloride (BMIC). Iron(II) chloride forms ionic liquids from a mole ratio of 1 FeCl(2)/3 BMIC to almost 1 FeCl(2)/1 BMIC. Both Raman scattering and ab initio calculations indicate that FeCl(4)(2-) is the predominant iron-containing species in these liquids. Iron(III) chloride forms ionic liquids from a mole ratio of 1 FeCl(3)/1.9 BMIC to 1.7 FeCl(3)/1 BMIC. When BMIC is in excess, Raman scattering indicates the presence of FeCl(4-). When FeCl(3) is in excess, Fe(2)Cl(7-) begins to appear and the amount of Fe(2)Cl(7-) increases with increasing amounts of FeCl(3). Ionic liquids were also prepared from a mixture of FeCl(2) and FeCl(3) and are discussed. Finally, we have used both Hartree-Fock and density functional theory methods to compute the optimized structures and vibrational spectra for these species. An analysis of the results using an all-electron basis set, 6-31G, as well as two different effective core potential basis sets, LANL2DZ and CEP-31G is presented.
Femtosecond lasers at fixed wavelengths above 1,000 nm are powerful, stable and inexpensive, making them promising sources for two-photon microscopy. Biosensors optimized for these wavelengths are needed for both next-generation microscopes and affordable turn-key systems. Here we report jYCaMP1, a yellow variant of the calcium indicator jGCaMP7 that outperforms its parent in mice and flies at excitation wavelengths above 1,000 nm and enables improved two-color calcium imaging with red fluorescent protein-based indicators.
Point-scanning two-photon microscopy enables high-resolution imaging within scattering specimens such as the mammalian brain, but sequential acquisition of voxels fundamentally limits imaging speed. We developed a two-photon imaging technique that scans lines of excitation across a focal plane at multiple angles and uses prior information to recover high-resolution images at over 1.4 billion voxels per second. Using a structural image as a prior for recording neural activity, we imaged visually-evoked and spontaneous glutamate release across hundreds of dendritic spines in mice at depths over 250 microns and frame-rates over 1 kHz. Dendritic glutamate transients in anaesthetized mice are synchronized within spatially-contiguous domains spanning tens of microns at frequencies ranging from 1-100 Hz. We demonstrate high-speed recording of acetylcholine and calcium sensors, 3D single-particle tracking, and imaging in densely-labeled cortex. Our method surpasses limits on the speed of raster-scanned imaging imposed by fluorescence lifetime.
The identification of active neurons and circuits in vivo is a fundamental challenge in understanding the neural basis of behavior. Genetically encoded calcium (Ca(2+)) indicators (GECIs) enable quantitative monitoring of cellular-resolution activity during behavior. However, such indicators require online monitoring within a limited field of view. Alternatively, post hoc staining of immediate early genes (IEGs) indicates highly active cells within the entire brain, albeit with poor temporal resolution. We designed a fluorescent sensor, CaMPARI, that combines the genetic targetability and quantitative link to neural activity of GECIs with the permanent, large-scale labeling of IEGs, allowing a temporally precise "activity snapshot" of a large tissue volume. CaMPARI undergoes efficient and irreversible green-to-red conversion only when elevated intracellular Ca(2+) and experimenter-controlled illumination coincide. We demonstrate the utility of CaMPARI in freely moving larvae of zebrafish and flies, and in head-fixed mice and adult flies.
A crystal structure of the anaerobic Ni-Fe-S carbon monoxide dehydrogenase (CODH) from Rhodospirillum rubrum has been determined to 2.8-Å resolution. The CODH family, for which the R. rubrum enzyme is the prototype, catalyzes the biological oxidation of CO at an unusual Ni-Fe-S cluster called the C-cluster. The Ni-Fe-S C-cluster contains a mononuclear site and a four-metal cubane. Surprisingly, anomalous dispersion data suggest that the mononuclear site contains Fe and not Ni, and the four-metal cubane has the form [NiFe3S4] and not [Fe4S4]. The mononuclear site and the four-metal cluster are bridged by means of Cys531 and one of the sulfides of the cube. CODH is organized as a dimer with a previously unidentified [Fe4S4] cluster bridging the two subunits. Each monomer is comprised of three domains: a helical domain at the N terminus, an α/β (Rossmann-like) domain in the middle, and an α/β (Rossmann-like) domain at the C terminus. The helical domain contributes ligands to the bridging [Fe4S4] cluster and another [Fe4S4] cluster, the B-cluster, which is involved in electron transfer. The two Rossmann domains contribute ligands to the active site C-cluster. This x-ray structure provides insight into the mechanism of biological CO oxidation and has broader significance for the roles of Ni and Fe in biological systems.
From insects to mammals, essential brain functions, such as forming long-term memories (LTMs), increase metabolic activity in stimulated neurons to meet the energetic demand associated with brain activation. However, while impairing neuronal metabolism limits brain performance, whether expanding the metabolic capacity of neurons boosts brain function remains poorly understood. Here, we show that LTM formation of flies and mice can be enhanced by increasing mitochondrial metabolism in central memory circuits. By knocking down the mitochondrial Ca exporter Letm1, we favour Ca retention in the mitochondrial matrix of neurons due to reduction of mitochondrial H/Ca exchange. The resulting increase in mitochondrial Ca over-activates mitochondrial metabolism in neurons of central memory circuits, leading to improved LTM storage in training paradigms in which wild-type counterparts of both species fail to remember. Our findings unveil an evolutionarily conserved mechanism that controls mitochondrial metabolism in neurons and indicate its involvement in shaping higher brain functions, such as LTM.
Genetically encoded calcium indicators (GECIs), together with modern microscopy, allow repeated activity measurement, in real time and with cellular resolution, of defined cellular populations. Recent efforts in protein engineering have yielded several high-quality GECIs that facilitate new applications in neuroscience. Here, we summarize recent progress in GECI design, optimization, and characterization, and provide guidelines for selecting the appropriate GECI for a given biological application. We focus on the unique challenges associated with imaging in behaving animals.
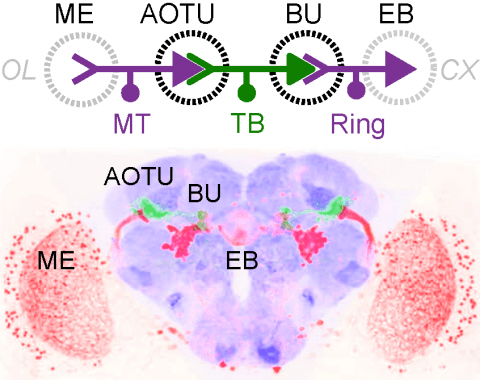
Many animals orient using visual cues, but how a single cue is selected from among many is poorly understood. Here we show that Drosophila ring neurons—central brain neurons implicated in navigation—display visual stimulus selection. Using in vivo two-color two-photon imaging with genetically encoded calcium indicators, we demonstrate that individual ring neurons inherit simple-cell-like receptive fields from their upstream partners. Stimuli in the contralateral visual field suppressed responses to ipsilateral stimuli in both populations. Suppression strength depended on when and where the contralateral stimulus was presented, an effect stronger in ring neurons than in their upstream inputs. This history-dependent effect on the temporal structure of visual responses, which was well modeled by a simple biphasic filter, may determine how visual references are selected for the fly's internal compass. Our approach highlights how two-color calcium imaging can help identify and localize the origins of sensory transformations across synaptically connected neural populations.
Metal ion homeostasis is critical to the survival of all cells. Regulation of nickel concentrations in Escherichia coli is mediated by the NikR repressor via nickel-induced transcriptional repression of the nickel ABC-type transporter, NikABCDE. Here, we report two crystal structures of nickel-activated E. coli NikR, the isolated repressor at 2.1 A resolution and in a complex with its operator DNA sequence from the nik promoter at 3.1 A resolution. Along with the previously published structure of apo-NikR, these structures allow us to evaluate functional proposals for how metal ions activate NikR, delineate the drastic conformational changes required for operator recognition, and describe the formation of a second metal-binding site in the presence of DNA. They also provide a rare set of structural views of a ligand-responsive transcription factor in the unbound, ligand-induced, and DNA-bound states, establishing a model system for the study of ligand-mediated effects on transcription factor function.