Filter
Result Type
Associated Lab
Associated Support Team
Tool Types
31 Results
Showing 1-10 of 31 resultsOrganismal aging involves functional declines in both somatic and reproductive tissues. Multiple strategies have been discovered to extend lifespan across species. However, how age-related molecular changes differ among various tissues and how those lifespan-extending strategies slow tissue aging in distinct manners remain unclear. Here we generated the transcriptomic Cell Atlas of Worm Aging (CAWA, http://mengwanglab.org/atlas ) of wild-type and long-lived strains. We discovered cell-specific, age-related molecular and functional signatures across all somatic and germ cell types. We developed transcriptomic aging clocks for different tissues and quantitatively determined how three different pro-longevity strategies slow tissue aging distinctively. Furthermore, through genome-wide profiling of alternative polyadenylation (APA) events in different tissues, we discovered cell-type-specific APA changes during aging and revealed how these changes are differentially affected by the pro-longevity strategies. Together, this study offers fundamental molecular insights into both somatic and reproductive aging and provides a valuable resource for in-depth understanding of the diversity of pro-longevity mechanisms.
Bacteria, omnipresent in our environment and coexisting within our body, exert dual beneficial and pathogenic influences. These microorganisms engage in intricate interactions with the human body, impacting both human health and disease. Simultaneously, certain organelles within our cells share an evolutionary relationship with bacteria, particularly mitochondria, best known for their energy production role and their dynamic interaction with each other and other organelles. In recent years, communication between bacteria and mitochondria has emerged as a new mechanism for regulating the host's physiology and pathology. In this review, we delve into the dynamic communications between bacteria and host mitochondria, shedding light on their collaborative regulation of host immune response, metabolism, aging, and longevity. Additionally, we discuss bacterial interactions with other organelles, including chloroplasts, lysosomes, and the endoplasmic reticulum (ER).
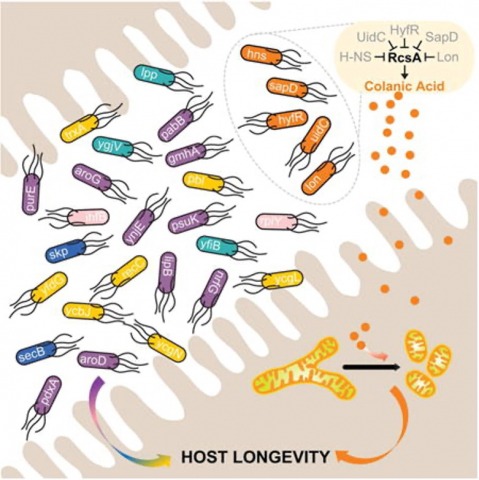
Microbiota-derived metabolites have emerged as key regulators of longevity. The metabolic activity of the gut microbiota, influenced by dietary components and ingested chemical compounds, profoundly impacts host fitness. While the benefits of dietary prebiotics are well-known, chemically targeting the gut microbiota to enhance host fitness remains largely unexplored. Here, we report a novel chemical approach to induce a pro-longevity bacterial metabolite in the host gut. We discovered that specific Escherichia coli strains overproduce colanic acids (CAs) when exposed to a low dose of cephaloridine, leading to an increased lifespan in host Caenorhabditis elegans. In the mouse gut, oral administration of low-dose cephaloridine induces the transcription of the capsular biosynthesis operon responsible for CA biosynthesis in commensal E. coli, which overcomes the inhibition of CA biosynthesis above 30 degrees C and enables its induction directly from the microbiota. Importantly, low-dose cephaloridine induces CA independently of its antibiotic properties through a previously unknown mechanism mediated by the membrane-bound histidine kinase ZraS. Our work lays the foundation for microbiota-based therapeutics through the chemical modulation of bacterial metabolism and reveals the promising potential of bacteria-targeting drugs in promoting host longevity.
Metabolism is fundamental to organism physiology and pathology. From the intricate network of metabolic reactions, diverse chemical molecules, collectively termed as metabolites, are produced. In multicellular organisms, metabolite communication between different tissues is vital for maintaining homeostasis and adaptation. However, the molecular mechanisms mediating these metabolite communications remain poorly understood. Here, we focus on nucleosides and nucleotides, essential metabolites involved in multiple cellular processes, and report the pivotal role of the SLC29A family of transporters in mediating nucleoside coordination between the soma and the germline. Through genetic analysis, we discovered that two Caenorhabditis elegans homologs of SLC29A transporters, Equilibrative Nucleoside Transporter ENT-1 and ENT-2, act in the germline and the intestine, respectively, to regulate reproduction. Their knockdown synergistically results in sterility. Further single-cell transcriptomic and targeted metabolomic profiling revealed that the ENT double knockdown specifically affects genes in the purine biosynthesis pathway and reduces the ratio of guanosine to adenosine levels. Importantly, guanosine supplementation into the body cavity/pseudocoelom through microinjection rescued the sterility caused by the ENT double knockdown, whereas adenosine microinjection had no effect. Together, these studies support guanosine as a rate limiting factor in the control of reproduction, uncover the previously unknown nucleoside/nucleotide communication between the soma and the germline essential for reproductive success, and highlight the significance of SLC-mediated cell-nonautonomous metabolite coordination in regulating organism physiology.
The scaffolding function of receptor interacting protein kinase 1 (RIPK1) confers intrinsic and extrinsic resistance to immune checkpoint blockades (ICBs) and emerges as a promising target for improving cancer immunotherapies. To address the challenge posed by a poorly defined binding pocket within the intermediate domain of RIPK1, here we harness proteolysis targeting chimera (PROTAC) technology to develop a RIPK1 degrader, LD4172. LD4172 exhibits potent and selective RIPK1 degradation both in vitro and in vivo. Degradation of RIPK1 by LD4172 triggers immunogenic cell death, enhances tumor-infiltrating lymphocyte responses, and sensitizes tumors to anti-PD1 therapy in female C57BL/6J mice. This work reports a RIPK1 degrader that serves as a chemical probe for investigating the scaffolding functions of RIPK1 and as a potential therapeutic agent to enhance tumor responses to ICBs therapy.