Filter
Associated Lab
- Aso Lab (30) Apply Aso Lab filter
- Betzig Lab (1) Apply Betzig Lab filter
- Bock Lab (2) Apply Bock Lab filter
- Branson Lab (9) Apply Branson Lab filter
- Card Lab (5) Apply Card Lab filter
- Clapham Lab (1) Apply Clapham Lab filter
- Dickson Lab (2) Apply Dickson Lab filter
- Druckmann Lab (1) Apply Druckmann Lab filter
- Fetter Lab (1) Apply Fetter Lab filter
- Funke Lab (2) Apply Funke Lab filter
- Harris Lab (3) Apply Harris Lab filter
- Heberlein Lab (2) Apply Heberlein Lab filter
- Hermundstad Lab (2) Apply Hermundstad Lab filter
- Hess Lab (6) Apply Hess Lab filter
- Jayaraman Lab (7) Apply Jayaraman Lab filter
- Lippincott-Schwartz Lab (1) Apply Lippincott-Schwartz Lab filter
- Looger Lab (2) Apply Looger Lab filter
- O'Shea Lab (1) Apply O'Shea Lab filter
- Otopalik Lab (1) Apply Otopalik Lab filter
- Reiser Lab (17) Apply Reiser Lab filter
- Riddiford Lab (1) Apply Riddiford Lab filter
- Romani Lab (2) Apply Romani Lab filter
- Rubin Lab (149) Apply Rubin Lab filter
- Saalfeld Lab (5) Apply Saalfeld Lab filter
- Scheffer Lab (8) Apply Scheffer Lab filter
- Schreiter Lab (1) Apply Schreiter Lab filter
- Simpson Lab (3) Apply Simpson Lab filter
- Singer Lab (1) Apply Singer Lab filter
- Spruston Lab (1) Apply Spruston Lab filter
- Stern Lab (1) Apply Stern Lab filter
- Svoboda Lab (3) Apply Svoboda Lab filter
- Tjian Lab (1) Apply Tjian Lab filter
- Truman Lab (4) Apply Truman Lab filter
- Turaga Lab (1) Apply Turaga Lab filter
- Turner Lab (5) Apply Turner Lab filter
- Zuker Lab (1) Apply Zuker Lab filter
Associated Project Team
- CellMap (1) Apply CellMap filter
- Fly Functional Connectome (4) Apply Fly Functional Connectome filter
- Fly Olympiad (3) Apply Fly Olympiad filter
- FlyEM (12) Apply FlyEM filter
- FlyLight (20) Apply FlyLight filter
- GENIE (1) Apply GENIE filter
- Transcription Imaging (1) Apply Transcription Imaging filter
Publication Date
- 2026 (2) Apply 2026 filter
- 2025 (8) Apply 2025 filter
- 2024 (4) Apply 2024 filter
- 2023 (5) Apply 2023 filter
- 2022 (1) Apply 2022 filter
- 2021 (4) Apply 2021 filter
- 2020 (9) Apply 2020 filter
- 2019 (6) Apply 2019 filter
- 2018 (7) Apply 2018 filter
- 2017 (15) Apply 2017 filter
- 2016 (3) Apply 2016 filter
- 2015 (16) Apply 2015 filter
- 2014 (9) Apply 2014 filter
- 2013 (5) Apply 2013 filter
- 2012 (8) Apply 2012 filter
- 2011 (4) Apply 2011 filter
- 2010 (4) Apply 2010 filter
- 2009 (2) Apply 2009 filter
- 2008 (4) Apply 2008 filter
- 2007 (2) Apply 2007 filter
- 2006 (1) Apply 2006 filter
- 2002 (1) Apply 2002 filter
- 2000 (2) Apply 2000 filter
- 1999 (1) Apply 1999 filter
- 1997 (1) Apply 1997 filter
- 1995 (2) Apply 1995 filter
- 1994 (2) Apply 1994 filter
- 1993 (2) Apply 1993 filter
- 1992 (1) Apply 1992 filter
- 1991 (2) Apply 1991 filter
- 1990 (3) Apply 1990 filter
- 1989 (2) Apply 1989 filter
- 1987 (2) Apply 1987 filter
- 1986 (1) Apply 1986 filter
- 1985 (1) Apply 1985 filter
- 1984 (1) Apply 1984 filter
- 1983 (1) Apply 1983 filter
- 1982 (2) Apply 1982 filter
- 1981 (1) Apply 1981 filter
- 1979 (1) Apply 1979 filter
- 1973 (1) Apply 1973 filter
Type of Publication
149 Publications
Showing 31-40 of 149 resultsThe behavioral state of an animal can dynamically modulate visual processing. In flies, the behavioral state is known to alter the temporal tuning of neurons that carry visual motion information into the central brain. However, where this modulation occurs and how it tunes the properties of this neural circuit are not well understood. Here, we show that the behavioral state alters the baseline activity levels and the temporal tuning of the first directionally selective neuron in the ON motion pathway (T4) as well as its primary input neurons (Mi1, Tm3, Mi4, Mi9). These effects are especially prominent in the inhibitory neuron Mi4, and we show that central octopaminergic neurons provide input to Mi4 and increase its excitability. We further show that octopamine neurons are required for sustained behavioral responses to fast-moving, but not slow-moving, visual stimuli in walking flies. These results indicate that behavioral-state modulation acts directly on the inputs to the directionally selective neurons and supports efficient neural coding of motion stimuli.
Analyzing Drosophila melanogaster neural expression patterns in thousands of three-dimensional image stacks of individual brains requires registering them into a canonical framework based on a fiducial reference of neuropil morphology. Given a target brain labeled with predefined landmarks, the BrainAligner program automatically finds the corresponding landmarks in a subject brain and maps it to the coordinate system of the target brain via a deformable warp. Using a neuropil marker (the antibody nc82) as a reference of the brain morphology and a target brain that is itself a statistical average of data for 295 brains, we achieved a registration accuracy of 2 μm on average, permitting assessment of stereotypy, potential connectivity and functional mapping of the adult fruit fly brain. We used BrainAligner to generate an image pattern atlas of 2954 registered brains containing 470 different expression patterns that cover all the major compartments of the fly brain.
Persistent internal states are important for maintaining survival-promoting behaviors, such as aggression. In female Drosophila melanogaster, we have previously shown that individually activating either aIPg or pC1d cell types can induce aggression. Here we investigate further the individual roles of these cholinergic, sexually dimorphic cell types, and the reciprocal connections between them, in generating a persistent aggressive internal state. We find that a brief 30-second optogenetic stimulation of aIPg neurons was sufficient to promote an aggressive internal state lasting at least 10 minutes, whereas similar stimulation of pC1d neurons did not. While we previously showed that stimulation of pC1e alone does not evoke aggression, persistent behavior could be promoted through simultaneous stimulation of pC1d and pC1e, suggesting an unexpected synergy of these cell types in establishing a persistent aggressive state. Neither aIPg nor pC1d show persistent neuronal activity themselves, implying that the persistent internal state is maintained by other mechanisms. Moreover, inactivation of pC1d did not significantly reduce aIPg-evoked persistent aggression, arguing that the aggressive state did not depend on pC1d-aIPg recurrent connectivity. Our results suggest the need for alternative models to explain persistent female aggression. Preprint: https://www.biorxiv.org/content/10.1101/2023.06.07.543722v2
Persistent internal states are important for maintaining survival-promoting behaviors, such as aggression. In female Drosophila melanogaster, we have previously shown that individually activating either aIPg or pC1d cell types can induce aggression. Here we investigate further the individual roles of these cholinergic, sexually dimorphic cell types, and the reciprocal connections between them, in generating a persistent aggressive internal state. We find that a brief 30-second optogenetic stimulation of aIPg neurons was sufficient to promote an aggressive internal state lasting at least 10 minutes, whereas similar stimulation of pC1d neurons did not. While we previously showed that stimulation of pC1e alone does not evoke aggression, persistent behavior could be promoted through simultaneous stimulation of pC1d and pC1e, suggesting an unexpected synergy of these cell types in establishing a persistent aggressive state. Neither aIPg nor pC1d show persistent neuronal activity themselves, implying that the persistent internal state is maintained by other mechanisms. Moreover, inactivation of pC1d did not significantly reduce aIPg-evoked persistent aggression arguing that the aggressive state did not depend on pC1d-aIPg recurrent connectivity. Our results suggest the need for alternative models to explain persistent female aggression.
The central complex (CX) plays a key role in many higher-order functions of the insect brain including navigation and activity regulation. Genetic tools for manipulating individual cell types, and knowledge of what neurotransmitters and neuromodulators they express, will be required to gain mechanistic understanding of how these functions are implemented. We generated and characterized split-GAL4 driver lines that express in individual or small subsets of about half of CX cell types. We surveyed neuropeptide and neuropeptide receptor expression in the central brain using fluorescent in situ hybridization. About half of the neuropeptides we examined were expressed in only a few cells, while the rest were expressed in dozens to hundreds of cells. Neuropeptide receptors were expressed more broadly and at lower levels. Using our GAL4 drivers to mark individual cell types, we found that 51 of the 85 CX cell types we examined expressed at least one neuropeptide and 21 expressed multiple neuropeptides. Surprisingly, all co-expressed a small neurotransmitter. Finally, we used our driver lines to identify CX cell types whose activation affects sleep, and identified other central brain cell types that link the circadian clock to the CX. The well-characterized genetic tools and information on neuropeptide and neurotransmitter expression we provide should enhance studies of the CX.
The central complex (CX) plays a key role in many higher-order functions of the insect brain including navigation and activity regulation. Genetic tools for manipulating individual cell types, and knowledge of what neurotransmitters and neuromodulators they express, will be required to gain mechanistic understanding of how these functions are implemented. We generated and characterized split-GAL4 driver lines that express in individual or small subsets of about half of CX cell types. We surveyed neuropeptide and neuropeptide receptor expression in the central brain using fluorescent in situ hybridization. About half of the neuropeptides we examined were expressed in only a few cells, while the rest were expressed in dozens to hundreds of cells. Neuropeptide receptors were expressed more broadly and at lower levels. Using our GAL4 drivers to mark individual cell types, we found that 51 of the 85 CX cell types we examined expressed at least one neuropeptide and 21 expressed multiple neuropeptides. Surprisingly, all co-expressed a small neurotransmitter. Finally, we used our driver lines to identify CX cell types whose activation affects sleep, and identified other central brain cell types that link the circadian clock to the CX. The well-characterized genetic tools and information on neuropeptide and neurotransmitter expression we provide should enhance studies of the CX.
Cellular ultrastructures for signal integration are unknown in any nervous system. The ellipsoid body (EB) of the Drosophila brain is thought to control locomotion upon integration of various modalities of sensory signals with the animal internal status. However, the expected excitatory and inhibitory input convergence that virtually all brain centres exhibit is not yet described in the EB. Based on the EB expression domains of genetic constructs from the choline acetyl transferase (Cha), glutamic acid decarboxylase (GAD) and tyrosine hydroxylase (TH) genes, we identified a new set of neurons with the characteristic ring-shaped morphology (R neurons) which are presumably cholinergic, in addition to the existing GABA-expressing neurons. The R1 morphological subtype is represented in the Cha- and TH-expressing classes. In addition, using transmission electron microscopy, we identified a novel type of synapse in the EB, which exhibits the precise array of two independent active zones over the same postsynaptic dendritic domain, that we named 'agora'. This array is compatible with a coincidence detector role, and represents ~8% of all EB synapses in Drosophila. Presumably excitatory R neurons contribute to coincident synapses. Functional silencing of EB neurons by driving genetically tetanus toxin expression either reduces walking speed or alters movement orientation depending on the targeted R neuron subset, thus revealing functional specialisations in the EB for locomotion control.
Aggressive social interactions are used to compete for limited resources and are regulated by complex sensory cues and the organism's internal state. While both sexes exhibit aggression, its neuronal underpinnings are understudied in females. Here, we identify a population of sexually dimorphic aIPg neurons in the adult central brain whose optogenetic activation increased, and genetic inactivation reduced, female aggression. Analysis of GAL4 lines identified in an unbiased screen for increased female chasing behavior revealed the involvement of another sexually dimorphic neuron, pC1d, and implicated aIPg and pC1d neurons as core nodes regulating female aggression. Connectomic analysis demonstrated that aIPg neurons and pC1d are interconnected and suggest that aIPg neurons may exert part of their effect by gating the flow of visual information to descending neurons. Our work reveals important regulatory components of the neuronal circuitry that underlies female aggressive social interactions and provides tools for their manipulation.
The study of synaptic specificity and plasticity in the CNS is limited by the inability to efficiently visualize synapses in identified neurons using light microscopy. Here, we describe synaptic tagging with recombination (STaR), a method for labeling endogenous presynaptic and postsynaptic proteins in a cell-type-specific fashion. We modified genomic loci encoding synaptic proteins within bacterial artificial chromosomes such that these proteins, expressed at endogenous levels and with normal spatiotemporal patterns, were labeled in an inducible fashion in specific neurons through targeted expression of site-specific recombinases. Within the Drosophila visual system, the number and distribution of synapses correlate with electron microscopy studies. Using two different recombination systems, presynaptic and postsynaptic specializations of synaptic pairs can be colabeled. STaR also allows synapses within the CNS to be studied in live animals noninvasively. In principle, STaR can be adapted to the mammalian nervous system.
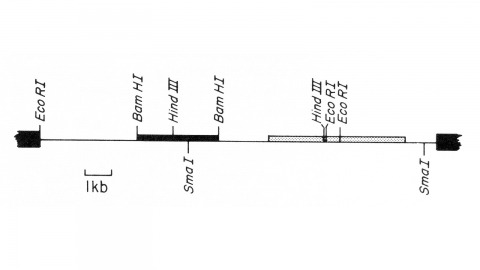
We describe the isolation of a cloned DNA segment carrying unique sequences from the white locus of Drosophila melanogaster. Sequences within the cloned segment are shown to hybridize in situ to the white locus region on the polytene chromosomes of both wild-type strains and strains carrying chromosomal rearrangements whose breakpoints bracket the white locus. We further show that two small deficiency mutations, deleting white locus genetic elements but not those of complementation groups contiguous to white, delete the genomic sequences corresponding to a portion of the cloned segment. The strategy we have employed to isolate this cloned segment exploits the existence of an allele at the white locus containing a copy of a previously cloned transposable, reiterated DNA sequence element. We describe a simple, rapid method for retrieving cloned segments carrying a copy of the transposable element together with contiguous sequences corresponding to this allele. The strategy described is potentially general and we discuss its application to the cloning of the DNA sequences of other genes in Drosophila, including those identified only by genetic analysis and for which no RNA product is known.